Scientific background
Structural data is a must for understanding the molecular basis for catalysis – Today, novel enzymes are sought and desired for their unique properties. Their catalytic properties are harnessed for a specific application, such as the degradation of biomass for biofuels, processing raw material for the food industry or as catalysts to perform novel chemical reactions as highlighted by the Nobel prize given to Frances Arnold in 2018.
However, most enzymes typically do not fulfil all process requirements and will need further fine-tuning to adapt to the strict requirement presented by an industrial-scale production. Typical inadequacies presented of the wild-type enzymes include low stability, substrate and/or product inhibition, low turnover rate and a limited substrate spectrum. These inherent properties of the initially discovered enzymes limit the immediate and practical application of the enzyme under investigation. To overcome these challenges, various tools of protein engineering have been developed, and researchers are now able to redesign the enzyme characteristics and tailor them for specific reactions. A common approach to enzyme design is to use the three‐dimensional crystal structure and apply a rational approach to systematically alter specific residues at key positions in the enzyme either in, or around the active site or at other interfacial regions required for function. This approach requires detailed knowledge from accumulated basic fundamental research describing both the structure and that the catalytic function of the enzyme, to make the desired and rational choices that induce functional changes. This knowledge is often unavailable for a vast majority of newly discovered enzymes. The lack of available crystals structures has been a major drawback for the use of rational design, especially since it earlier could take over a year to determine a crystal structure.
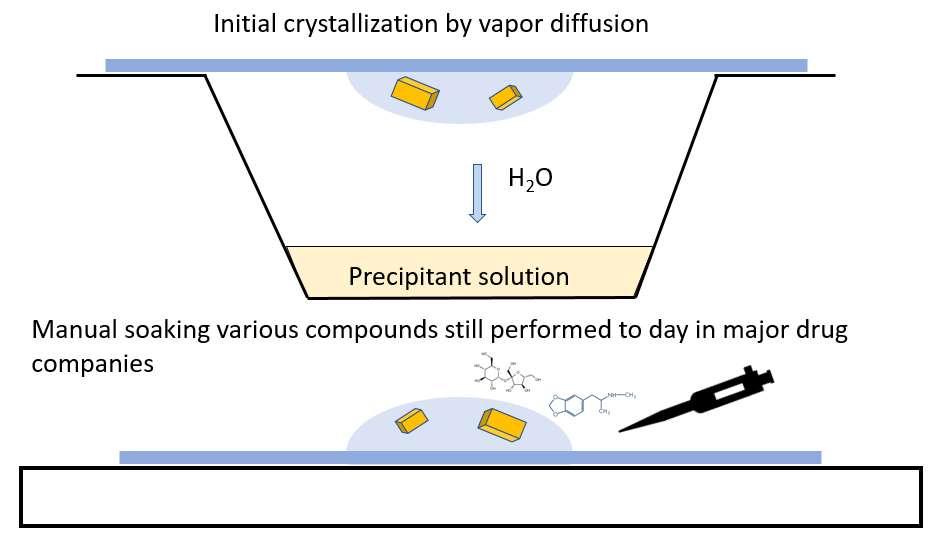
Crystallization
Crystallization – A technique used for more than just structure determination. Crystallization of macromolecules and structure determination using X-rays have become a mainstream technique, and remain the most powerful technique to obtain atomic resolution molecular structures of any chemical molecules. Crystals of macromolecules typically include large solvent channels as part of the crystal lattice, this advantage is specific to macromolecular crystallization, and allows external compounds to access the individual macromolecules while still stabilized by the crystal lattice. This makes macromolecular crystals formidable workbenches to explore for ligand binding, metal exchange or drug discovery.